
Understanding the Long-Term Effects of Opioid Use on Body and Mind
Over the past two decades, the dramatic increase in opioid prescriptions has resulted in a significant rise in their adverse health effects. Often prescribed for
Hydrocodone is an opiate agonist, semi-synthetic narcotic analgesic, and antitussive (cough suppressant) used to treat mild to moderate pain. This drug is synthesized from one of the alkaloids found in the opium poppy, codeine. In fact, the International Narcotics Control Boards report that 99% of its consumption is in the United States. Due to the opioid crisis, the administration has changed its classification from Schedule III to Schedule II. This means doctors can no longer prescribe HCPs by phone, and patients must present a written prescription to the pharmacist at the time of purchase. Besides, hydrocodone-based drugs have been linked to many overdoses and ER admissions in the last few years.
Speak Confidentially
with an opiate detox treatment expert.
Immediate-release with paracetamol:
Immediate-release with ibuprofen:
Immediate-release with aspirin:
Controlled-release:
CDC Reports: Deaths from prescription opioids—drugs like oxycodone, hydrocodone, and methadone—have more than quadrupled.
Long-term use of opioids such as Hydrocodone has been proven to possibly lessen the pain-relieving effects (Hyperalgesia) because the body develops a dependence on the drug. Thus, this dependence causes several withdrawal symptoms, making people fearful of stopping their use. In particular, addiction occurs when physical dependence interferes with your life quality.
For example, typical side effects can include:

You should seek immediate emergency medical help if you have any of these symptoms because they could be the signs of a severe allergic reaction.
For example, other symptoms that require you to call your doctor are:

To illustrate, overdose symptoms include cold/clammy skin, circulatory collapse, stupor, coma, depression, respiratory depression, cardiac arrest, and death. Furthermore, mixing hydrocodone with other substances, including alcohol, can cause severe physical problems or death. Therefore, understanding the distinction between symptoms and signs of hydrocodone abuse can save lives. Hydrocodone is an opioid drug that can be incredibly addictive. On the other hand, friends and family members who can recognize addiction signs can intervene and offer immediate help.
CDC Reports: Deaths from prescription opioids—drugs like oxycodone, hydrocodone, and methadone—have more than quadrupled.
Hydrocodone addiction often includes uncontrollable cravings for the drug and the inability to control the use. Additionally, the necessity to use and acquire drugs will trump and adverse effects that drug use might be having on your health, personal relationships, or finances. Hence, people who are addicted to hydrocodone may vow to quit, and they may have every intention to do so. However, making the right decisions does not come easy anymore because the body and mind have been altered. Also, people are scared of suffering from withdrawal symptoms and failing to promise others. Therefore, it is essential to acknowledge that whether someone develops a mild, moderate, or severe opioid use disorder, effective treatment is available and complete recovery is possible.
The first stage of recovery is overcoming the physical aspects of withdrawal, specifically detoxification. However, withdrawal symptoms from Hydrocodone can be highly unpleasant and challenging to overcome. The initial period of opioid detoxification can be intense, but the most successful way to get through it is with adequate medical support.
In general, an inpatient medical facility is always the safest choice, especially for patients using multiple drugs, over 40 years of age, or other health issues. On occasion, opioid withdrawal can lead to complications that might need immediate medical attention. For this reason, it is often not a good idea for people to attempt detox at home. Instead, enrollment at an inpatient medical detox provides patients with the ongoing medical monitoring and symptom management necessary to succeed.
Finding the right hydrocodone treatment program can be just a phone call away from you.
Call us today, and we will help you find the detox option that is right for you.
We have over 22 years of experience, and we are here to help you.
Speak With an Addiction Specialist
Exceptional Care & Better Outcome. Get In Touch With Us Today!
What's New at Waismann Method®

Over the past two decades, the dramatic increase in opioid prescriptions has resulted in a significant rise in their adverse health effects. Often prescribed for

Pain management is a critical component of healthcare, particularly when addressing conditions that involve both pain and inflammation. Two commonly used medications in this regard
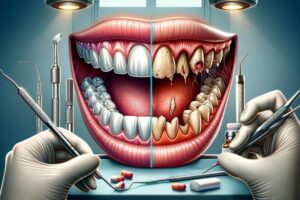
Opioids serve as potent pain relievers in medical settings, but their recreational use presents serious risks, including a variety of dental health problems. Whether prescribed
We are available 7 days a week
Get Newsletter Updates from Waismann Method®
"*" indicates required fields
DISCLAIMER: The text presented on this page is not a substitute for professional medical advice. It is for your information only and may not represent your true individual medical situation. Do not hesitate to consult your healthcare provider if you have any questions or concerns. Do not use this information to diagnose or treat a health problem or disease without consulting a qualified healthcare professional. Be advised that Opiates.com articles are derived from various sources and may not reflect your own country’s regulations.