
Understanding the Long-Term Effects of Opioid Use on Body and Mind
Over the past two decades, the dramatic increase in opioid prescriptions has resulted in a significant rise in their adverse health effects. Often prescribed for
Opioid addiction is a serious problem that affects millions of people worldwide. It can lead to a number of health problems, including overdose, addiction, and death. Buprenorphine is a safe medication that helps people in high risk, prevent withdrawal, while controlling their addiction.
Buprenorphine is a partial opioid agonist, which means that it binds to the same receptors in the brain as opioids like heroin and morphine. However, buprenorphine produces a weaker effect than these opioids, which reduces the risk of overdose and addiction.
This medication is available in a number of different formulations, including:
Buprenorphine is also available in combination with other medications, such as naloxone. Naloxone is a medication that blocks the effects of opioids. It is added to some buprenorphine formulations to help prevent overdose.
Speak Confidentially
with an opiate detox treatment expert.
Buprenorphine mechanism of action is quite unique and interesting. Here’s a breakdown of how it works:
Long-Acting: Buprenorphine is long-acting, meaning it stays in the body for an extended period. This property is beneficial for treating opioid addiction as it allows for dosing once a day or even less frequently, which is more convenient and can help with compliance to the treatment regimen.
Sublingual and Transdermal Forms: It’s typically administered sublingually (under the tongue) or via transdermal patches. The sublingual form ensures that the medication is absorbed directly into the bloodstream, bypassing the digestive system where it could be broken down and become less effective.
Combination with Naloxone: Often, buprenorphine is combined with naloxone to form a medication known as Suboxone. Naloxone is an opioid antagonist that blocks the effects of opioids. It is included in the formulation to deter misuse; if Suboxone is injected, the naloxone will block the effects of buprenorphine and precipitate withdrawal symptoms, but when taken as directed sublingually, naloxone has negligible effects.
In summary, the distinct pharmacological characteristics of buprenorphine render it a safer choice for individuals grappling with opioid addiction, particularly amid the escalating fentanyl crisis. Its unique action as a partial opioid agonist, combined with a high affinity for mu-opioid receptors and a ceiling effect on its use, significantly lowers the risk of abuse and dangerous side effects, offering a more secure treatment alternative in these challenging times.
Benefits of Buprenorphine Maintenance Therapy
Clinical research has consistently demonstrated the benefits of buprenorphine maintenance therapy in treating opioid addiction. It has been shown to:
While buprenorphine is a valuable tool for treating opioid addiction, it is important to acknowledge its potential for dependence. Like other opioids, buprenorphine can lead to physical dependence, meaning the body adapts to its presence and experiences withdrawal symptoms when it is discontinued. However, the risk of developing a severe addiction to buprenorphine is significantly lower than that of other opioids.
Buprenorphine maintenance therapy should be initiated and supervised by a qualified healthcare provider who specializes in addiction treatment. This provider can assess the individual’s needs, determine the appropriate dosage, and monitor progress throughout the treatment course.
At our hospital, we provide a specialized buprenorphine detoxification process under sedation for individuals preparing to discontinue buprenorphine medications. This approach is particularly beneficial for those who find it challenging to safely undergo detoxification or manage buprenorphine withdrawal symptoms at home, ensuring a smoother and more successful detox experience.
Learn more about Buprenorphine detox and precautions.
Speak With an Addiction Specialist
Exceptional Care & Better Outcome. Get In Touch With Us Today!
At the Waismann Method, we are dedicated to providing an advanced and highly effective approach to buprenorphine detoxification. Our program is a testament to our commitment to innovation and excellence in addiction treatment.
Embarking on recovery with the Waismann Method means choosing a life of renewed health and well-being. Our rapid detox is more than a treatment; it’s a transformative experience that empowers you to reclaim control over your life.
Contact us today at 1-800-423-2482 to discover how the Waismann Method can be the cornerstone of your journey toward a life free of opioid dependence. Our team is ready to support you every step of the way.
Resources about buprenorphine for opioid addiction treatment:
What's New at Waismann Method®

Over the past two decades, the dramatic increase in opioid prescriptions has resulted in a significant rise in their adverse health effects. Often prescribed for

Pain management is a critical component of healthcare, particularly when addressing conditions that involve both pain and inflammation. Two commonly used medications in this regard
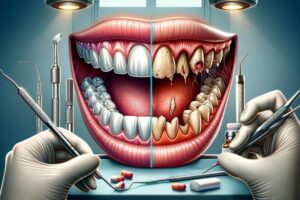
Opioids serve as potent pain relievers in medical settings, but their recreational use presents serious risks, including a variety of dental health problems. Whether prescribed
We are available 7 days a week
Get Newsletter Updates from Waismann Method®
"*" indicates required fields
DISCLAIMER: The text presented on this page is not a substitute for professional medical advice. It is for your information only and may not represent your true individual medical situation. Do not hesitate to consult your healthcare provider if you have any questions or concerns. Do not use this information to diagnose or treat a health problem or disease without consulting a qualified healthcare professional. Be advised that Opiates.com articles are derived from various sources and may not reflect your own country’s regulations.